1. Introduction
On May 21, 2010, the 63rd World Assembly of the World Health Organization adopted a resolution that established a World Hepatitis Day on July 28, and stated that “This endorsement by member states calls for WHO to develop a comprehensive approach to the prevention and control of these diseases.” The diseases were the viral hepatitides A through E. This resolution, and a second one relating to alcoholic liver disease, represent the first formal declaration by WHO that the burden of liver disease represents a major global public health problem. However, although viral hepatitis and alcoholic liver disease are critical to global health, they do not encompass all—or even the most important—of the conditions contributing to the global health burden due to liver diseases. Over the past couple of decades, it has become increasingly clear that nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH) are now the number one cause of liver disease in Western countries. The prevalence of NAFLD has doubled during last 20 years, whereas the prevalence of other chronic liver diseases has remained stable or even decreased. More recent data confirm that NAFLD and NASH play an equally important role in the Middle East, Far East, Africa, the Caribbean, and Latin America.
NAFLD is a condition defined by excessive fat accumulation in the form of triglycerides (steatosis) in the liver (> 5% of hepatocytes histologically). A subgroup of NAFLD patients have liver cell injury and inflammation in addition to excessive fat (steatohepatitis). The latter condition, designated NASH, is virtually indistinguishable histologically from alcoholic steatohepatitis (ASH). While the simple steatosis seen in NAFLD does not correlate with increased short-term morbidity or mortality, progression of this condition to that of NASH dramatically increases the risks of cirrhosis, liver failure, and hepatocellular carcinoma (HCC). Cirrhosis due to NASH is an increasingly frequent reason for liver transplantation. While the morbidity and mortality from liver causes are greatly increased in patients with NASH, they correlate even more strongly with the morbidity and mortality from cardiovascular disease.
Table 1 Mortality in NAFLD/NASH

NASH is widely considered to be the liver expression of the metabolic syndrome—diseases related to diabetes mellitus type 2, insulin resistance, central (truncal) obesity, hyperlipidemia (low high-density lipoprotein cholesterol, hypertriglyceridemia), and hypertension. There is at present a worldwide epidemic of diabetes and obesity. At least 1.46 billion adults were overweight or obese and 170 million of the world’s children were overweight or obese in 2008. In some parts of Africa, obesity afflicts more children than malnutrition. The numbers are continuing to rise, indicating that NASH will become an increasingly common liver problem in both rich and poor countries, increasing the global burden of liver disease and affecting public health and health-care costs globally. It is estimated that NAFLD/NASH will increase 5-year direct and indirect medical costs by 26%.
Table 2 Clinical identification of the metabolic syndrome (scientific statement by the American Heart Association and National Heart, Lung, and Blood Institute in the United States)

HDL, high-density lipoprotein.
The exact cause of NASH has not been elucidated, and it is almost certainly not the same in every patient. Although it is most closely related to insulin resistance, obesity, and the metabolic syndrome, not all patients with these conditions have NAFLD/NASH, and not all patients with NAFLD/NASH suffer from one of these conditions. However, as noted above, NASH is a potentially fatal condition, leading to cirrhosis, liver failure, and HCC.
There is no established therapy and there are no evidence-based clinical guidelines. There have not been any adequate prospective, double-blind, controlled trials to provide the data necessary to create an evidence-based guideline. This Global Guideline is intended to provide the best opinions of a group of experts from all areas of the globe concerning every aspect of this problem and the best approaches to diagnosing and treating this condition, taking locally available resources into account.
Cascades—a resource-sensitive approach
A gold standard approach is feasible for regions and countries in which the full scale of diagnostic tests and medical treatment options are available for the management of NASH. However, such resources are not available throughout much of the world. With their diagnostic and treatment cascades, the World Gastroenterology Organisation guidelines provide a resource-sensitive approach.
Cascade: a hierarchical set of diagnostic, therapeutic, and management options to deal with risk and disease, ranked by the resources available.
2. Epidemiology
NASH is an increasingly common chronic liver disease with worldwide distribution that is closely associated with diabetes and obesity, which have both reached epidemic proportions. It is estimated that there are at least 1.46 billion obese adults worldwide. Approximately 6 million individuals in the USA are estimated to have progressed to NASH and some 600,000 to NASH-related cirrhosis. There are significant cultural and geographic differences in the prevalence of obesity.
Whereas in most Western countries, the preferred body image, especially in women, is very thin with minimal body fat, that is not necessarily true globally. In many other cultures, obesity is considered desirable and also regarded as a distinct sign of prosperity (see, for example, the data from Egypt given below).
In the USA, obesity is particularly epidemic in those from lower socio-economic groups who rely heavily on diets provided by high-fat, high-calorie fast food outlets (“junk food”). The opposite is true in many poorer countries, where it is the well-todo, better-educated population that has the highest prevalence of obesity.
Regional obesity/overweight data
Table 3 Regional obesity/overweight data (representative examples)

BMI, body mass index.
Fig. 1 Estimated prevalence of obesity (BMI > 25) in males and females aged 15+ (2010).

Source: WHO InfoBase.
Table 4 Overweight and obesity—summary of prevalence by region (2004)

Source: WHO 2009 [25]. Click here to link to the source.
Prevalence of NAFLD and NASH
Table 5 Estimated prevalences of NAFLD and NASH. Reports on the prevalence of NAFLD and NASH vary substantially due to varying definitions, differences in the populations studied, and the diagnostic methods used

3. Pathogenesis and risk factors
NASH represents the most severe histologic form of nonalcoholic fatty liver disease (NAFLD), which is defined by fat accumulation in the liver exceeding 5% of its weight. Uniform criteria for diagnosing and staging NASH are still debated (see details in later sections).
Insulin resistance is related to obesity and is central to the pathogenesis of NAFLD. In addition, oxidative stress and cytokines are important contributing factors, together resulting in steatosis and progressive liver damage in genetically susceptible individuals.
Key histologic components of NASH are steatosis, hepatocellular ballooning, and lobular inflammation; fibrosis is not part of the histologic definition of NASH. However, the degree of fibrosis on liver biopsy (stage) is predictive of the prognosis, whereas the degree of inflammation and necrosis on liver biopsy (grade) are not.
The disease can remain asymptomatic for years, or can progress to cirrhosis and hepatocellular carcinoma.
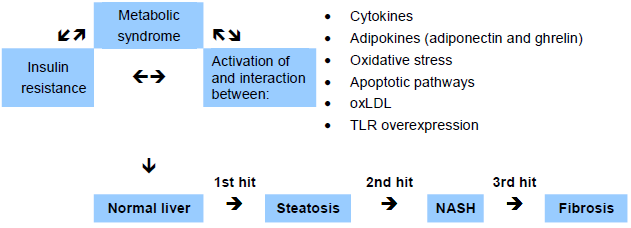
One global hypothesis for the pathogenesis of NASH is the “multi-hit hypothesis,” with metabolic syndrome playing a major role, due to insulin resistance and the proinflammatory process mediated by different proteins and immune components. The identities of the multiple “hits” are different in each patient and largely undefined at present.
Fig. 2 The “multi-hit” hypothesis for nonalcoholic steatohepatitis (NASH). oxLDL, oxidized low-density lipoprotein; TLR, Toll-like receptor.
Risk factors and associated conditions
The characteristics of a low-risk population are: young, healthy, with low alcohol consumption, and not obese.
Table 6 Risk factors and associated conditions

Table 7 Calculation of insulin resistance

HOMA, homeostasis model assessment; QUICKI, quantitative insulin-sensitivity check index.
Table 8 NASH scoring system in morbid obesity

Prognosis and complications
- Disease progression from NAFLD to NASH to cirrhosis/liver failure and HCC.
- NAFLD does not exacerbate hepatotoxicity, and side effects of pharmacologic agents, including HMG-CoA reductase inhibitors, are not more likely to occur,
- NAFLD and coexistent obesity and related metabolic factors may exacerbate other liver diseases—e.g., alcoholic liver disease.
- Concurrence of NAFLD with hepatitis C or human immunodeficiency virus (HIV) worsens their prognoses and decreases their responses to therapy.
- Hepatitis C, genotype 3, is commonly associated with hepatic steatosis, which may confuse a diagnosis of hepatitis C vs. NASH vs. both together.
- Liver biopsy may indicate the severity of disease, but only fibrosis, and not inflammation or necrosis, has been confirmed to predict the disease prognosis.
- Histologic progression to end-stage liver disease may occur: NASH + bridging fibrosis or cirrhosis.
- End-stage NASH is an often under-recognized cause of cryptogenic cirrhosis; progressive fibrosis may be obscured by stable or improving steatosis and serologic features, especially in older NASH patients.
- NASH-related (cryptogenic) cirrhosis increases the risk of hepatocellular carcinoma (HCC).
- Causes of mortality in cirrhotic NASH patients:
- Liver failure
- Sepsis
- Variceal hemorrhage
- HCC
- Cardiovascular disease
Table 9 NASH survival rates in comparison with simple steatosis and alcoholic steatohepatitis (ASH)

Table 10 Disease progression from NAFLD to NASH to cirrhosis/liver failure and HCC. The results of prevalence and incidence studies vary substantially due to varying definitions, different populations studied, and diagnostic methods used

- Independent predictors for progression of fibrosis:
- Age > 45–50
- BMI > 28–30 kg/m2
- Degree of insulin resistance
- Diabetes
- Hypertension
- Negative impact on NASH survival:
- Diabetes and elevated serum alanine (ALT) and aspartate aminotransferase (AST)
- Older age and presence of necrotic inflammation on initial liver biopsy
- Older age, impaired fasting glucose, and presence of cirrhosis
4. Diagnosis
Patient history and clinical evaluation
- Patient symptoms:
- In most cases, NASH does not cause any specific symptoms.
- There are sometimes vague symptoms of fatigue, malaise, and abdominal discomfort.
- The presence of any of the following, especially with a history of abnormal AST/ALT, should lead to a work-up for NAFLD/NASH:
- Presence of obesity, especially morbid obesity (BMI > 35)
- Diagnosis of type 2 diabetes mellitus
- Diagnosis of metabolic syndrome
- History of obstructive sleep apnea
- Presence of insulin resistance (see below and Table 7)
- Chronic elevation of AST/ALT, otherwise unexplained
- Detailed patient history of alcohol consumption—threshold < 20 g/day in women, < 30 g/day in men. This is critical, as no diagnostic test can reliably distinguish between ASH and NASH.
- Appropriate specialized questionnaires or scoring systems for the evaluation of alcohol consumption should be used.
- CAGE questionnaire: CAGE is the acronym for the four questions: have you ever felt you needed to Cut down on your drinking, that people Annoyed you by criticizing your drinking, felt Guilty about drinking, needed a drink first thing in the morning (Eye-opener)? CAGE is a widely used method of screening for alcoholism, and confirms clinically relevant alcohol consumption if at least one of the questions is answered positively and if the Alcohol Use Disorders Identification Test (AUDIT) score is higher than 8.
- Although it is generally recommended that one should avoid all alcohol if one has underlying liver disease, this can raise problems in patients with the metabolic syndrome who have documented coronary artery disease, for whom modest wine consumption has been shown to be beneficial. Limited studies have suggested that modest wine drinking (0.12 L / 4 fluid ounces per day) may be associated with a decreased prevalence of NAFLD. Its effectiveness as treatment for preexisting NAFLD has not been addressed.
- Central obesity correlates with severity of inflammation on biopsy, and dorsocervical lipohypertrophy (buffalo hump) correlates with hepatocyte injury.
- Physical findings in case of progression/advanced liver disease: spider angiomas, ascites, hepatomegaly, splenomegaly, palmar erythema, jaundice, hepatic encephalopathy.
Routine laboratory findings and imaging tests
- Elevated ALT and AST:
- In 10% of NASH patients, ALT and AST may be normal, especially with simple steatosis.
- An abnormal ferritin level in the presence of normal transferrin saturation should always suggest a need to rule out NASH.
- AST/ALT ratio < 1—this ratio is usually > 2 in alcoholic hepatitis.
- Typical imaging test results confirming fat accumulation in the liver:
- The magnetic resonance imaging (MRI) test has a quantitative value, but cannot distinguish between NASH and ASH.
- Ultrasound is the usual screening test for fatty liver.
No imaging study can identify fat accurately if it is < 33% or distinguish NASH from ASH.
Tests to exclude:
- Viral hepatitis—hepatitis B surface antigen, hepatitis C virus antibody or HCVRNA, hepatitis A antibody IgM, hepatitis E antibody (in an appropriate geographical setting); it should be noted that the patient may have coexisting viral hepatitis as well as NAFLD/NASH.
- Alcohol-related liver disease including alcoholic steatohepatitis.
- Autoimmune liver disease.
- Congenital causes of chronic liver disease: hereditary hemochromatosis, Wilson’s disease, alpha-1-antitrypsin deficiency, polycystic ovary syndrome.
- Drug-induced liver disease.
Investigational laboratory tests, scoring systems, and imaging modalities
A wide variety of attempts have been made to develop scoring systems or imaging techniques that will allow noninvasive diagnosis of NASH and avoid the need for a liver biopsy. Currently, none has been tested rigorously enough in prospective, double-blind studies, nor has their ability to predict the prognosis or response to therapy been proven. The majority of speciality serum tests/scores are available from single laboratories or research laboratories and only at significant cost, so they are of little value in countries with limited resources. Specialized imaging modalities, including FibroScan, using a novel “controlled attenuation parameter,” and positron emission tomography (PET) scanning suffer from the same limitations of limited availability, high cost, and lack of sufficient controlled data.
An extensive review of the various modalities and the data currently available can be found in the article by Dowman et al. [7]. Another detailed discussion of the issues was published in Ratziu et al. [11]. The methods involved show great promise for the future, but cannot at present be recommended at this time for general use.
Liver biopsy
Although it is invasive and has a potential for sampling errors and inconsistent interpretation of the histopathology, liver biopsy is required in order to establish the diagnosis and to stage NASH. The currently most commonly used histological scoring system is summarized in Table 11. It is used primarily in controlled trials to evaluate the effects of experimental therapies, rather than to establish the diagnosis of NASH. It has been independently validated and is applicable to both adult and pediatric NAFLD/NASH. There is no reliable way of distinguishing between NAFLD/ALD and NASH/ASH without a liver biopsy. Because of the difficulties in proper interpretation of the liver biopsy, it is best if it can be read by a specialized hepatopathologist with experience in making the histopathologic diagnosis.
Table 11 NASH Clinical Research Network histological scoring system

Source: Kleiner et al., Hepatology 2005;41:1313–21 [35].
Liver biopsy and histology are indicated in order to confirm a NASH diagnosis, to grade and stage the disease, and to rule out other diagnoses in the presence of one or more of the following findings:
- Abnormal serum ferritin in the absence of an elevated transferrin saturation
- Cytopenia
- Splenomegaly
- Clinical signs of chronic liver disease
- Diabetes and abnormal persistently elevated AST/ALT
- Obesity and age > 45 or abnormal AST/ALT
- Unexplained hepatomegaly
Table 12 Diagnostic tests for fatty liver

ALT, alanine aminotransferase; ASH, alcoholic steatohepatitis; AST, aspartate aminotransferase; CT, computed tomography; MRI, magnetic resonance imaging; MRS, magnetic resonance spectroscopy; NASH, nonalcoholic steatohepatitis.
Diagnostic strategy for NASH
Fig. 3 Management algorithm for NAFLD. Based on Rafiq and Younossi [10].

Liver enzyme tests and liver ultrasound:
- In patients who seek medical help in relation to insulin resistance/metabolic syndrome/diabetes
Imaging procedures to evaluate for steatosis:
- In patients with elevated liver enzymes
Liver biopsy:
- May be indicated if there is a strong suspicion for advanced fibrosis, when liver enzymes are elevated and ultrasound is positive for steatosis.
- To determine the severity of disease/fibrosis when noninvasive tests are indeterminate.
- Indicated in patients with chronic liver disease (other than NAFLD) and positive tests for metabolic risk factors, insulin resistance, and steatosis on ultrasound.
- If elevated ferritin with normal transferrin saturation, must rule out NASH.
- During surgical procedures in other high-risk groups—e.g., anti-obesity surgery, cholecystectomy.
None of the noninvasive tests will rule out other possible underlying diseases or stage the disease for prognostic purposes.
Ultimately, NAFLD/NASH is a diagnosis of exclusion, and liver biopsy will often be required to confirm the diagnosis, stage the disease, rule out other liver diseases, and determine the need for and urgency of aggressive therapy.
Fig. 4 Algorithm for liver biopsy in patients with suspected NAFLD after exclusion of other liver diseases.

Fig. 5 Diagnostic options for NAFLD

ALT, alanine aminotransferase; AMA, antimitochondrial antibody; ANA, antinuclear antibody; anti-LKM Ab, anti-liver–kidney microsomal antibody; ASMA, anti-smooth muscle antibody; AST, aspartate aminotransferase; BMI, body mass index; CT, computed tomography; FBG, fasting blood glucose; GGT, gamma-glutamyl transferase; HBsAg, hepatitis B surface antigen; HCV, hepatitis C virus; LFT, liver function tests; OGTT, oral glucose tolerance test.
Cascade—options for diagnosis in patients with suspected NAFLD/NASH
Table 13 Diagnostic cascade for extensive, medium, and limited resources

Ab, antibody; HbA1c, glycosylated hemoglobin; HBsAg, hepatitis B surface antigen; HCV, hepatitis C virus; HEV, hepatitis E virus; MRI, magnetic resonance imaging.

Ab, antibody; HbA1c, glycosylated hemoglobin; HBsAg, hepatitis B surface antigen; HCV, hepatitis C virus; HEV, hepatitis E virus.

5. Management
Therapeutic rationale
Targets for therapy are insulin resistance and oxidative stress. Although several treatment options are being evaluated, the value of most treatments remains uncertain, or the effects reverse when they are discontinued. The goals of treatment for NASH are to reduce the histologic features and improve insulin resistance and liver enzyme levels.
At the present time, there is no evidence-based approved drug therapy for NAFLD/NASH. Lifestyle change is critical in any attempt to reverse the course of NAFLD/NASH.
In the absence of a treatment that would represent a standard of care, the management of NASH focuses on associated conditions. NASH should be treated aggressively in order to prevent progression to cirrhosis, as these patients are frequently not candidates for liver transplantation due to their morbid obesity, cardiovascular disease, or other complications of their underlying condition.
The overall goal of lifestyle change is to reduce excess weight: even a gradual 5–10% weight loss has been shown to improve liver histology and enzymes, but not fibrosis. This is usually most successful if combined with an active exercise program and elimination of a sedentary lifestyle. This may also require a sensitive approach to explaining the problems of obesity in certain cultures in which it may be considered a mark of beauty/desirability and/or prosperity.
Liver transplantation is appropriate in the face of liver failure. Some 30–40% of patients with NASH-related cirrhosis require liver transplantation. Most programs will decline patients with an elevated BMI (which varies from > 35 to > 45, depending on local program criteria). NASH can recur in the transplanted liver, or a new occurrence may even develop.
Treatment options for NASH
As emphasized above, lifestyle changes are critical in any attempt to reverse the course of NAFLD/NASH, and an evidence-based approved drug therapy for NAFLD/NASH is not available at present.
Treatment of metabolic conditions
Proper control of diabetes, hyperlipidemia, and cardiovascular risks is recommended. Studies with atorvastatin and pravastatin have shown improvement in histology in patients with NASH. NAFLD patients with dyslipidemia should be treated with statins. Patients with underlying liver disease do not seem to have any additional risk of statin toxicity. Serious hepatotoxicity from statins is rare.
Improving insulin sensitivity—weight reduction
- Diet: A weight loss of 5–10% should be aimed for, and a 25% decrease in calories from the normal diet (ca. 2500 calories per day) for the patient’s age and sex. A moderately calorie-restricted diet with modified macronutrient composition produces better results in comparison with a very low-caloric diet. Attention should be given to the role of a hypocaloric diet and counseling about the type of foods to be consumed—avoiding fructose and trans-fats present in soft drinks and fast foods, and increasing omega-3/omega-6 polyunsaturated fatty acids in diet. This may be difficult for the patient to adhere to, and many patients regain weight after an initial loss.
- Exercise: A moderate exercise program three to four times a week should be encouraged to achieve a heart rate of 60–75% of the age-based maximum.
- The efficacy of dietary and exercise measures should be assessed after a 6-month period; if they have been ineffective, additional therapeutic options such as pharmacologic therapy may then be considered.
- Weight loss (bariatric) surgery may be beneficial for patients with morbid obesity; again, this should be considered early, as most programs will decline such surgery for patients who are already cirrhotic. Limited studies have reported a dramatic improvement in liver disease, as well as other complications of metabolic syndrome/insulin resistance, following successful bariatric surgery.
- Drugs targeting insulin resistance, such as thiazolidinediones and metformin, are approved for diabetes therapy but not for NAFLD/NASH, and should be considered experimental (see the reference list below for more information and detailed discussion).
Antioxidants and antifibrotic agents
Antioxidants and antifibrotic agents, such as vitamin E and pentoxifylline, have not been approved for NASH/NAFLD treatment. For all of them, there are limited data and few if any data from double-blind controlled trials. They are all considered experimental (see the reference list below for more information and detailed discussion).
Monitoring strategy
Disease progression and complications can be detected during the follow-up as indicated in Table 14.
Table 14 Follow-up tests and their timing

Cascades—options for therapy
Table 15 Therapy cascades for extensive, medium, and limited resources



6. Summary
- NAFLD and NASH represent a major global public health problem, which is pandemic and affects rich and poor countries alike.
- There is insufficient evidence to justify screening for NASH/advanced liver disease in the general population.
- The diagnosis should be sought in all patients who present with risk factors for NASH. Not all patients with risk factors will have NAFLD or NASH, and not all patients with NASH will have standard risk factors.
- Not every person with fatty liver needs aggressive therapy.
- Diet and exercise should be instituted for all patients.
- Liver biopsy should be reserved for those patients who have risk factors for NASH and/or other liver diseases.
- Patients with NASH or risk factors for NASH should first be treated with diet and exercise. Vitamin E or pentoxifylline may be added in these patients. Experimental therapy should be considered only in appropriate hands and only in patients who fail to achieve a 5–10% weight reduction over 6 months–1 year of successful lifestyle changes.
- Bariatric surgery should be considered in patients in whom the above approaches fail, and it should be performed before the patient becomes cirrhotic.
- Liver transplantation is successful in patients who meet the criteria for liver failure, but NASH may recur after transplantation and is likely to be denied to patients with morbid obesity.
- NAFLD and NASH are also becoming an increasingly serious problem in pediatric patients, including those under the age of 10.
- Ultimately, NAFLD and NASH are diagnoses of exclusion and require careful consideration of other diagnoses. Just as the clinician cannot diagnose NASH on the basis of clinical data alone, the pathologist can document the histological lesions of steatohepatitis, but cannot reliably distinguish those of nonalcoholic origin from those of alcoholic origin.
References
Position statements and reviews
Insufficient randomized, controlled, double blind studies are available to provide evidence-based data for a formal guideline, as discussed in the Introduction above. The following is a listing of selected position statements, reviews, and expert opinion articles.
- Angulo P. Nonalcoholic fatty liver disease. N Engl J Med 2002;346:1221–31. PMID: 11961152.
- Angulo P. Diagnosing steatohepatitis and predicting liver-related mortality in patients with NAFLD: two distinct concepts. Hepatology 2011;53:1792–4. doi: 10.1002/hep.24403. PMID: 21557278.
- Brunt EM. Nonalcoholic steatohepatitis. Semin Liver Dis 2004;24:3–20. PMID: 15085483.
- Chalasani N, Younossi Z, Lavine JE, Diehl AM, Brunt EM, Cusi K, et al. The diagnosis and management of non-alcoholic fatty liver disease: Practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012;55:2005–23. doi: 10.1002/hep.25762. PMID: 22488764
- Cheung O, Sanyal AJ. Recent advances in nonalcoholic fatty liver disease. Curr Opin Gastroenterol 2010;26:202–8. PMID: 20168226.
- Clark JM, Brancati FL, Diehl AM. Nonalcoholic fatty liver disease. Gastroenterology 2002;122:1649–57. PMID: 12016429.
- Dowman JK, Tomlinson JW, Newsome PN. Systematic review: the diagnosis and staging of non-alcoholic steatohepatitis. Aliment Pharmacol Ther 2011;33:525–40. doi:10.1111/j.1365- 2036-2010.04556.x. Epub 2010 Dec 29. PMID: 21198708.
- Fabbrini E, Sullivan S, Klein S. Obesity and nonalcoholic fatty liver disease: biochemical, metabolic, and clinical implications. Hepatology 2010;51:679–89. PMID: 20041406.
- Lancet 2011 Aug 27–Sept 2;378(9793): virtually this entire issue addresses the global obesity pandemic, with articles on world epidemiology, cultural and political costs, pathogenesis, therapy, and proposed approaches to the problem. A virtual primer on global obesity. Articles are detailed in the next section, under Epidemiology.
- Rafiq N, Younossi ZM. Nonalcoholic fatty liver disease: a practical approach to evaluation and management. Clin Liver Dis 2009;13:249–66. PMID: 19442917.
- Ratziu V, Bellentani S, Cortez-Pinto H, Day C, Marchesini G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J Hepatol 2010;53:372–84. Epub 2010 May 7. PMID: 20494470
- Sanyal AJ, Brunt EM, Kleiner DE, Kowdley KV, Chalasani N, Lavine JE, et al. Endpoints and clinical trial design for nonalcoholic steatohepatitis. Hepatology 2011;54:344–53. doi: 10.1002/hep.24376. PMID: 21520200.
- Torres DM, Harrison SA. Diagnosis and therapy of nonalcoholic steatohepatitis. Gastroenterology 2008;134:1682–98. PMID: 18471547.
- Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther 2011;24:274–85. doi: 10.1111/j.1365-2036.2011.04724.x. Epub 2011 May 30. PMID: 2162852.
- Vuppalachi R, Chalasani N. Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: selected practical issues in their evaluation and management. Hepatology 2009;49:306–17. PMID: 19065650.
Further reading
For those wishing additional information and documentation of the basis for the recommendations given in this guideline, selected references are listed below under the headings of epidemiology, pediatric epidemiology, histologic diagnosis, noninvasive diagnosis, hepatitis C and NAFLD/NASH, pathophysiology, and treatment.
Epidemiology
- Lancet 2011 Aug 27–Sept 2;378(9793).
- Editorial. Urgently needed: a framework convention for obesity control Lancet 2011;378:742.PMID: 21872732.
- Baur LA. Changing perceptions of obesity—recollections of a paediatrician. Lancet 2011;378:762–3. PMID: 21877330.
- Dietz WH. Reversing the tide of obesity. Lancet 2011;378:744–6. PMID: 21872735.
- Freudenberg N. The social science of obesity. Lancet 2011;378:760.
- Gortmaker SL, Swinburn BA, Levy D, Carter R, Mabry PL, Finegood DT, et al. Changing the future of obesity: science, policy, and action. Lancet 2011;378:838–47. PMID: 21872752.
- Hall KD, Sacks G, Chandramohan D, Chow CC, Wang YC, Gortmaker SL, et al. Quantification of the effect of energy imbalance on bodyweight. Lancet 2011;378:826–37. PMID: 21872751.
- King D. The future challenge of obesity. Lancet 2011;378:743–4. PMID: 21872734.
- Mozaffarian D. Diets from around the world—quality not quantity. Lancet 2011;378:759.
- Pincock S. Boyd Swinburn: combating obesity at the community level. Lancet 2011;378:761. PMID: 21872738.
- Rutter H. Where next for obesity? Lancet 2011;378:746–7. PMID: 21872736.
- Swinburn BA, Sacks G, Hall KD, McPherson K, Finegood DT, Moodie ML, et al. The global obesity pandemic: shaped by global drivers and local environments. Lancet 2011;378:804–14.
- Wang YC, McPherson K, Marsh T, Gortmaker SL, Brown M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet 2011;378:815–25. PMID: 21872750.
- Adams LA. Mortality in nonalcoholic fatty liver disease: clues from the Cremona study. Hepatology 2011;54:6–8. doi: 10.1002/hep.24445. PMID: 21618568.
- Centers for Disease Control and Prevention. 1990–2010 changes of percentage of obese adults in the USA (BMI > 30). Available at: www.cdc.gov/obesity/data/trends.html.
- Danaei G, Finucane MM, Lu Y, Singh GM, Cowan MJ, Paciorek CJ, et al. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet 2011;378:31–40. Epub 2011 Jun 24. PMID: 21705069.
- Gastaldelli A, Kozakova M, Højlund K, Flyvbjerg A, Favuzzi A, Mitrakou A, et al. Fatty liver is associated with insulin resistance, risk of coronary heart disease, and early atherosclerosis in a large European population. Hepatology 2009;49:1537–44. PMID: 19291789.
- Gu D, Reynolds K, Wu X, Chen J, Duan X, Reynolds RF, et al. Prevalence of the metabolic syndrome and overweight among adults in China. Lancet 2005;365:1398–405.
- Ludwig DS, Currie J. The association between pregnancy weight gain and birthweight: a withinfamily comparison. Lancet 2010;376:984–90. Epub 2010 Aug 4. PMID: 20691469.
- Passas G, Akhtar T, Gergen P, Hadden WC, Kahn AQ. Health status of the Pakistani population: a health profile and comparison with the United States. Am J Public Health 2001;91:93–8.
- Williams CD, Stengel J, Asike MI, Torres DM, Shaw J, Contreras M, et al. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: a prospective study. Gastroenterology 2011;140:124–31. Epub 2010 Sep 19. PMID: 20858492.
- World Health Organization. Global health risks: mortality and burden of disease attributable to selected major risks. Geneva: World Health Organization, 2009; Overweight and obesity—summary of prevalence by region. Geneva: World Health Organization, 2004. Available at: www.who.int/evidence/bod and www.who.int/healthinfo/global_burden_disease/risk_factors/en/index.html.
- Younossi ZM, Stepanova M, Afendy M, Fang Y, Younossi Y, Mir H, et al. Changes in the prevalence of the most common causes of chronic liver diseases in the United States from 1988 to 2008. Clin Gastroenterol Hepatol 2011;9:524–530.e1; quiz e60. Epub 2011 Mar 25. PMID:21440669.
Pediatric epidemiology
- Alkhouri N, Carter-Kent C, Lopez R, Rosenberg WM, Pinzani M, Bedogni G, et al. A combination of the pediatric NAFLD fibrosis index and enhanced liver fibrosis test identifies children with fibrosis. Clin Gastroenterol Hepatol 2011;9:150–5. Epub 2010 Oct 1. PMID: 20888433.
- Galal OM. The nutrition transition in Egypt: obesity, undernutrition and the food consumption context. Public Health Nutr 2002;5:141–8. Review. PMID: 12027277.
- Kerkar N. Non-alcoholic steatohepatitis in children. Pediatr Transplant 2004;8:613–8. PMID: 15598336.
- Mathur P, Das MK, Arora NK. Non-alcoholic fatty liver disease and childhood obesity. Indian J Pediatr 2007;74:401–7. PMID: 17476088.
- Salazar-Martinez E, Allen B, Fernandez-Ortega C, Torres-Mejia G, Galal O, Lazcano-Ponce E. Overweight and obesity status among adolescents from Mexico and Egypt. Arch Med Res 2006;37:535–42. PMID: 16624655.
Histologic diagnosis
- Angulo P. Long-term mortality in nonalcoholic fatty liver disease: is liver histology of any prognostic significance? Hepatology 2010;51:373–5. Erratum in: Hepatology 2010 May;51(5):1868. PMID: 20101746.
- Brunt EM, Tiniakos DG. Histopathology of nonalcoholic fatty liver disease. World J Gastroenterol 2010;16:5286–96. Review. PMID: 21072891.
- Brunt EM, Janney CG, Di Bisceglie AM, Neuschwander-Tetri BA, Bacon BR. Nonalcoholic steatohepatitis: a proposal for grading and staging the histological lesions. Am J Gastroenterol 1999;94:2467–74. PMID: 10484010.
- Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005;41:1313–21. PMID: 15915461.
- Tiniakos DG. Nonalcoholic fatty liver disease/nonalcoholic steatohepatitis: histological diagnostic criteria and scoring systems. Eur J Gastroenterol Hepatol 2010;22:643–50. PMID: 19478676.
- Younossi ZM, Stepanova M, Rafiq N, Makhlouf H, Younoszai Z, Agrawal R, et al. Pathologic criteria for nonalcoholic steatohepatitis: interprotocol agreement and ability to predict liverrelated mortality. Hepatology 2011;53:737–45. doi: 10.1002/hep.24131. Epub 2011 Feb 11. PMID: 21360720.
Noninvasive diagnosis
- Angulo P, Hui JM, Marchesini G, Bugianesi E, George J, Farrell GC, et al. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology 2007;45:846–54. PMID: 17393509.
- Babor TF, Higgins-Biddle JC, Saunders JB, Monteiro MG. The alcohol use disorders identification test: guidelines for use in primary care. 2nd ed. Geneva: World Health Organization, 2001.
- Baranova A, Younossi ZM. The future is around the corner: noninvasive diagnosis of progressive nonalcoholic steatohepatitis. Hepatology 2008;47:373–5. PMID: 18220279.
- Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, Castiglione A, et al. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol 2006;6:33. PMID: 17081293.
- Calori G, Lattuada G, Ragogna F, Garancini MP, Crosignani P, Villa M, et al. Fatty liver index and mortality: the Cremona study in the 15th year of follow-up. Hepatology 2011;54:145–52. doi: 10.1002/hep.24356. PMID: 21488080.
- Campos GM, Bambha K, Vittinghoff E, Rabl C, Posselt AM, Ciovica R, et al. A clinical scoring system for predicting nonalcoholic steatohepatitis in morbidly obese patients. Hepatology 2008;47:1916–23. PMID: 18433022.
- Chalasani N. Nonalcoholic fatty liver disease liver fat score and fat equation to predict and quantitate hepatic steatosis: promising but not prime time! Gastroenterology 2009;137:772–5. Epub 2009 Jul 26. PMID: 19638269.
- Cho CS, Curran S, Schwartz LH, Kooby DA, Klimstra DS, Shia J, et al. Preoperative radiographic assessment of hepatic steatosis with histologic correlation. J Am Coll Surg 2008;206:480–8. Epub 2007 Nov 26. PMID: 18308219.
- Hernaez R, Lazo M, Bonekamp S, Kamel I, Brancati FL, Guallar E, et al. Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: A meta-analysis. Hepatology 2011;54:1082–9. doi: 10.1002/hep.24452. PMID: 21618575.
- Hettihawa LM, Palangasinghe S, Jayasinghe SS, Gunasekara SW, Weerarathna TP. Comparison of insulin resistance by indirect methods—HOMA, QUICKI and McAuley—with fasting insulin in patients with type 2 diabetes in Galle, Sri Lanka: a pilot study. Online J Health Allied Sci 2006;1:2. Available at: http://www.ojhas.org/issue17/2006-1-2.htm.
- Hrebícek J, Janout V, Malincíková J, Horáková D, Cízek L. Detection of insulin resistance by simple quantitative insulin sensitivity check index QUICKI for epidemiological assessment and prevention. J Clin Endocrinol Metab 2002;87:144–7. PMID: 11788638.
- Imbert-Bismut F, Naveau S, Morra R, Munteanu M, Ratziu V, Abella A, et al. The diagnostic value of combining carbohydrate-deficient transferrin, fibrosis, and steatosis biomarkers for the prediction of excessive alcohol consumption. Eur J Gastroenterol Hepatol 2009;21:18–27. PMID: 19011575.
- Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, Sullivan G, et al. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab 2000;85:2402–10. PMID: 10902758.
- Keskin M, Kurtoglu S, Kendirci M, Atabek ME, Yazici C. Homeostasis model assessment is more reliable than the fasting glucose/insulin ratio and quantitative insulin sensitivity check index for assessing insulin resistance among obese children and adolescents. Pediatrics 2005;115:e500–3. Epub 2005 Mar 1. PMID: 15741351.
- Kotronen A, Peltonen M, Hakkarainen A, Sevastianova K, Bergholm R, Johansson LM, et al. Prediction of non-alcoholic fatty liver disease and liver fat using metabolic and genetic factors. Gastroenterology 2009;137:865–72. Epub 2009 Jun 12. PMID: 19524579.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412–9. PMID: 3899825.
- Poynard T, Ratziu V, Naveau S, Thabut D, Charlotte F, Messous D, et al. The diagnostic value of biomarkers (SteatoTest) for the prediction of liver steatosis. Comp Hepatol 2005;4:10. PMID: 16375767.
- Saadeh S, Younossi ZM, Remer EM, Gramlich T, Ong JP, Hurley M, et al. The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 2002;123:745–50. PMID: 12198701.
- Stern SE, Williams K, Ferrannini E, DeFronzo RA, Bogardus C, Stern MP. Identification of individuals with insulin resistance using routine clinical measurements. Diabetes 2005;54:333–9. PMID: 15677489.
- Thabut D, Naveau S, Charlotte F, Massard J, Ratziu V, Imbert-Bismut F, et al. The diagnostic value of biomarkers (AshTest) for the prediction of alcoholic steato-hepatitis in patients with chronic alcoholic liver disease. J Hepatol 2006;44:1175–85. Epub 2006 Mar 13. PMID: 16580087.
- Yajima Y, Ohta K, Narui T, Abe R, Suzuki H, Ohtsuki M. Ultrasonographic diagnosis of fatty liver: significance of the liver–kidney contrast. Tohoku J Exp Med 1983;139:43–50. PMID: 6220488.
Hepatitis C and NAFLD/NASH
- Bugianesi E, Marchesini G, Gentilcore E, Cua IH, Vanni E, Rizzetto M, et al. Fibrosis in genotype 3 chronic hepatitis C and nonalcoholic fatty liver disease: role of insulin resistance and hepatic steatosis. Hepatology 2006;44:1648–55. PMID: 17133473.
- Charlton MR, Pockros PJ, Harrison SA. Impact of obesity on treatment of chronic hepatitis C. Hepatology 2006;43:1177–86. PMID: 16729327.
- Everhart JE, Lok AS, Kim HY, Morgan TR, Lindsay KL, Chung RT, et al. Weight-related effects on disease progression in the hepatitis C antiviral long-term treatment against cirrhosis trial. Gastroenterology 2009;137:549–57. Epub 2009 May 13. PMID: 19445938.
- Koike K. Hepatitis C as a metabolic disease: Implication for the pathogenesis of NASH. Hepatol Res 2005;33:145–50. Epub 2005 Oct 3. PMID: 16202646.
- Negro F, Clément S. Impact of obesity, steatosis and insulin resistance on progression and response to therapy of hepatitis C. J Viral Hepat 2009;16:681–8. Epub 2009 Sep 1. PMID: 19732324.
- Popkin BM. Is the obesity epidemic a national security issue around the globe? Curr Opin Endocrinol Diabetes Obes 2001;18:328–31. PMID: 21543976.
- Powell EE, Jonsson JR, Clouston AD. Metabolic factors and non-alcoholic fatty liver disease as co-factors in other liver diseases. Dig Dis 2010;28:186–91. Epub 2010 May 7. PMID: 20460909.
- Sanyal AJ, Banas C, Sargeant C, Luketic VA, Sterling RK, Stravitz RT, et al. Similarities and differences in outcomes of cirrhosis due to nonalcoholic steatohepatitis and hepatitis C. Hepatology 2006;43:682–9. PMID: 16502396.
- Targher G, Day CP, Bonora E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med 2010;363:1341–50. PMID: 20879883.
Pathophysiology
- Ascha MS, Hanouneh IA, Lopez R, Tamimi TA, Feldstein AF, Zein NN. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology 2010;51:1972–8. PMID: 20209604.
- de Alwis NM, Day CP. Non-alcoholic fatty liver disease: the mist gradually clears. J Hepatol 2008;48(Suppl 1):S104–12. Epub 2008 Feb 4. PMID: 18304679.
- Lim JS, Mietus-Snyder M, Valente A, Schwarz JM, Lustig RH. The role of fructose in the pathogenesis of NAFLD and the metabolic syndrome. Nat Rev Gastroenterol Hepatol 2010;7:251–64. Epub 2010 Apr 6. PMID: 20368739.
- Neuschwander-Tetri BA. Evolving pathophysiologic concepts in nonalcoholic steatohepatitis. Am J Gastroenterol 2001;96:2813–4. PMID: 11693313.
- Polyzos SA, Kountouras J, Zavos C. The multi-hit process and the antagonistic role of tumor necrosis factor-alpha and adiponectin in non alcoholic fatty liver disease. Hippokratia 2009;13:127. PMID:19561788.
- Zein CO, Unalp A, Colvin R, Liu YC, McCullough AJ; Nonalcoholic Steatohepatitis Clinical Research Network. Smoking and severity of hepatic fibrosis in nonalcoholic fatty liver disease. J Hepatol 2011;54:753–9. Epub 2010 Sep 22. PMID: 21126792.
Treatment
- Abdelmalek MF, Suzuki A, Guy C, Unalp-Arida A, Colvin R, Johnson RJ, et al. Increased fructose consumption is associated with fibrosis severity in patients with nonalcoholic fatty liver disease. Hepatology 2010;51:1961–71. PMID: 20301112.
- Brunt EM, Kleiner DE, Wilson LA, Belt P, Neuschwander-Tetri BA; NASH Clinical Research Network (CRN). Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: distinct clinicopathologic meanings. Hepatology 2011;53:810–20. doi: 10.1002/hep.24127. Epub 2011 Feb 11. PMID: 21319198
- Bugianesi E, Gentilcore E, Manini R, Natale S, Vanni E, Villanova N, et al. A randomized controlled trial of metformin versus vitamin E or prescriptive diet in nonalcoholic fatty liver disease. Am J Gastroenterol 2005;100:1082–90. PMID: 15842582.
- Charlton MR, Burns JM, Pedersen RA, Watt KD, Heimbach JK, Dierkhising RA. Frequency and outcomes of liver transplantation for nonalcoholic steatohepatitis in the United States. Gastroenterology 2011;141:1249–53. Epub 2011 Jul 2. PMID: 21726509.
- Chavez-Tapia NC, Tellez-Avila FL, Barrientos-Gutierrez T, Mendez-Sanchez N, Lizardi- Cervera J, Uribe M. Bariatric surgery for non-alcoholic steatohepatitis in obese patients. Cochrane Database Syst Rev 2010;(1):CD007340. PMID: 20091629.
- Dunn W, Xu R, Schwimmer JB. Modest wine drinking and decreased prevalence of suspected nonalcoholic fatty liver disease. Hepatology 2008;47:1947–54. PMID: 18454505.
- Geier A. Shedding new light on vitamin D and fatty liver disease. J Hepatol 2011;55:273–5. Epub 2011 Jan 12. PMID: 21236303.
- Georgescu EF. Angiotensin receptor blockers in the treatment of NASH/NAFLD: could they be a first-class option? Adv Ther 2008;25:1141–74. PMID: 18972077.
- Henriksen JH, Ring-Larsen H. Rosiglitazone: possible complications and treatment of nonalcoholic steatohepatitis (NASH). J Hepatol 2008;48:174–6. Epub 2007 Nov 5. PMID: 18022724.
- Mummadi RR, Kasturi KS, Chennareddygari S, Sood GK. Effect of bariatric surgery on nonalcoholic fatty liver disease: systematic review and meta-analysis. Clin Gastroenterol Hepatol 2008;6:1396–402. Epub 2008 Aug 19. PMID: 18986848.
- Nakano T, Cheng YF, Lai CY, Hsu LW, Chang YC, Deng JY, et al. Impact of artificial sunlight therapy on the progress of non-alcoholic fatty liver disease in rats. J Hepatol 2011;55:415–25. Epub 2010 Dec 22. PMID: 21184788
- Neuschwander-Tetri BA. NASH: Thiazolidinediones for NASH—one pill doesn’t fix everything. Nat Rev Gastroenterol Hepatol 2010;7:243–4. PMID: 20442730.
- Promrat K, Kleiner DE, Niemeier HM, Jackvony E, Kearns M, Wands JR, et al. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010;51:121–9. PMID: 19827166.
- Ratziu V, Giral P, Jacqueminet S, Charlotte F, Hartemann-Heutier A, Serfaty L, et al. Rosiglitazone for nonalcoholic steatohepatitis: one-year results of the randomized placebocontrolled Fatty Liver Improvement with Rosiglitazone Therapy (FLIRT) Trial. Gastroenterology 2008;135:100–10. Epub 2008 Apr 8. PMID: 18503774
- Sanyal AJ, Chalasani N, Kowdley KV, McCullough A, Diehl AM, Bass NM, et al. Pioglitazone, vitamine E, or placebo for nonalcoholic steatohepatitis. N Engl J Med 2010;362:1675–85. Epub 2010 Apr 28. PMID: 20427778.
- Suzuki A, Lindor K, St. Saver J, Lymp J, Mendes F, Muto A, et al. Effect of changes on body weight and lifestyle in nonalcoholic fatty liver disease. J Hepatol 2005;43:1060–6. Epub 2005 Jul 11. PMID: 16140415.
- Zein CO, Yerian LM, Gogate P, Lopez R, Kirwan JP, Feldstein AE, et al. Pentoxifylline improves nonalcoholic steatohepatitis: a randomized placebo-controlled trial. Hepatology 2011;54:1610–9. doi: 10.1002/hep.24544. Epub 2011 Aug 24. PMID: 21748765.
- Zelber-Sagi S, Nitzan-Kaluski D, Goldsmith R, Webb M, Zvibel I, Goldiner I, et al. Role of leisure-time physical activity in nonalcoholic fatty liver disease: a population-based study. Hepatology 2008;48:1791–8. PMID: 18972405.




























